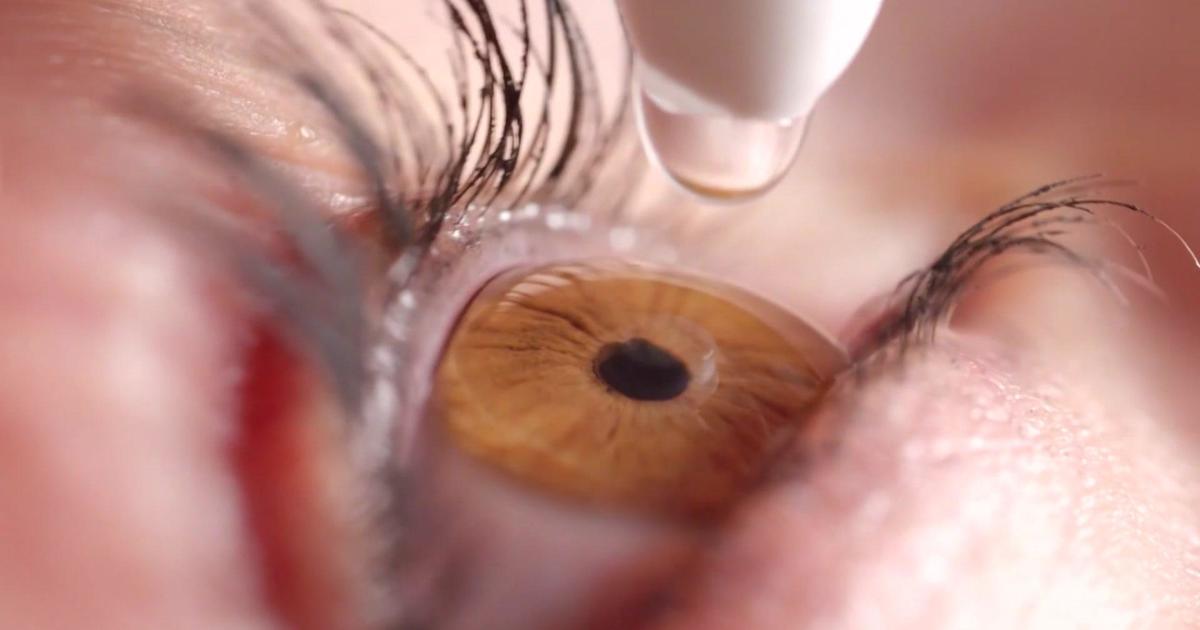
FDA warns of contaminated copycat eye drops
CBSN
The Food and Drug Administration is warning the public against copycat eye drops because of the risk of infection.
South Moon, Rebright and FivFivGo eye drops are offered in packaging that could easily be mistaken for Bausch + Lomb's Limify brand eye drops, an over-the-counter product approved for redness relief, the FDA said on Wednesday.
The copycat products claim to address conditions like glaucoma, which is treated with prescription drugs or surgery, the agency noted.

On the day that marks 13 years since the death of Venezuelan socialist strongman Hugo Chávez and two months after the Jan. 3 U.S. operation that captured Nicolás Maduro, the scene in Caracas looks strikingly different from the anti-U.S.-imperialism rhetoric that founded Chavismo and was echoed by his successor. In:

Defense Secretary Pete Hegseth deemed artificial intelligence firm Anthropic a "supply chain risk to national security" on Friday, following days of increasingly heated public conflict over the company's effort to place guardrails on the Pentagon's use of its technology. Jo Ling Kent contributed to this report. In:







