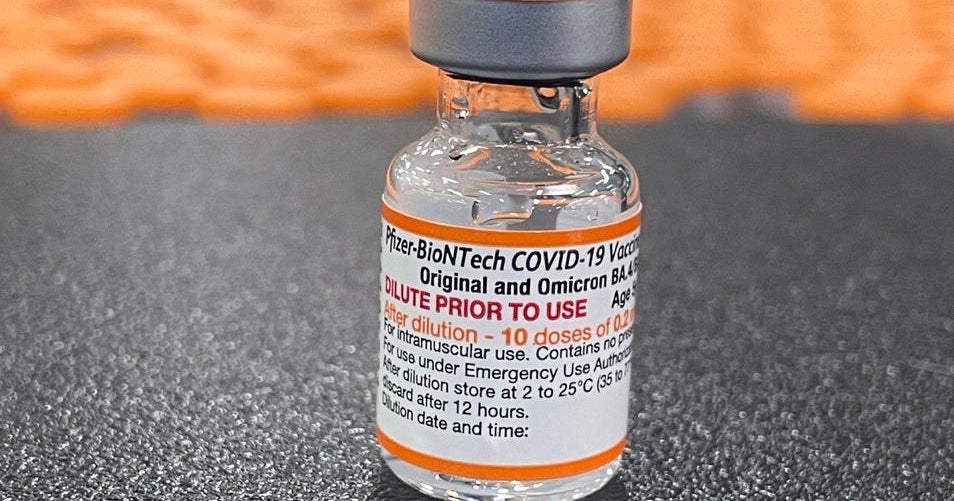
FDA clears updated COVID boosters for kids as young as 5 years old
CBSN
The Food and Drug Administration announced Wednesday that it had granted emergency use authorization for updated COVID-19 vaccine boosters for Americans as young as 5 years old. The boosters can be given at least two months after their previous shot of the vaccine.
The Centers for Disease Control and Prevention is expected to sign off on the shots soon, clearing the final hurdle before eligibility for the bivalent vaccines can be expanded to these younger age groups.
A CDC spokesperson said the agency's outside vaccine advisers have "already reviewed the relevant data" at a previous meeting and would not need to be reconvened, "in order to avert delays" in expanding eligibility for the shots.

A jury has found Elon Musk liable for misleading investors by deliberately driving down Twitter's stock price in the tumultuous months leading up to his 2022 acquisition of the social media company for $44 billion. But it absolved him of some fraud allegations, finding that he did not "scheme" to mislead investors. In:












