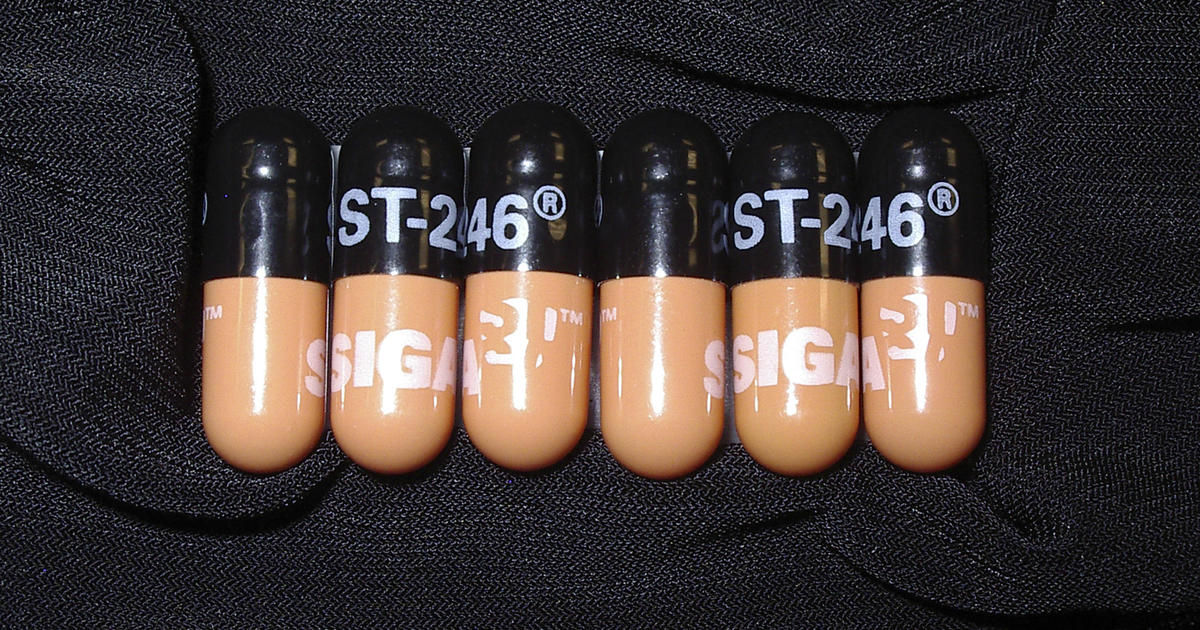
FDA warns monkeypox could mutate if antiviral drug is overused
CBSN
The monkeypox virus is only one mutation away from evading a key antiviral drug being used to treat at-risk patients, federal health officials are now warning — and they're urging doctors to be "judicious" in prescribing the sought-after treatment.
The new FDA guidance for the antiviral drug known as tecovirimat, or Tpoxx, was published this week online and in updated labeling.
The regulator says lab and animal studies, and evidence from a human case of this family of viruses, suggest monkeypox has "several genetic pathways" to evolve resistance to tecovirimat. Many "require only a single amino acid change," the FDA said.

A jury on Wednesday found that Meta and YouTube are liable for creating products that led to harmful and addictive behavior by young users, a landmark decision that could set a legal precedent for similar allegations brought against social media companies. Edited by Alain Sherter and Aimee Picchi In:

An internal watchdog report in the Department of Homeland Security identified serious vulnerabilities in TSA's screenings at airports nationwide — and the agency has yet to respond five months later, according to internal communications provided to House Homeland Security Committee staff and reviewed by CBS News.











