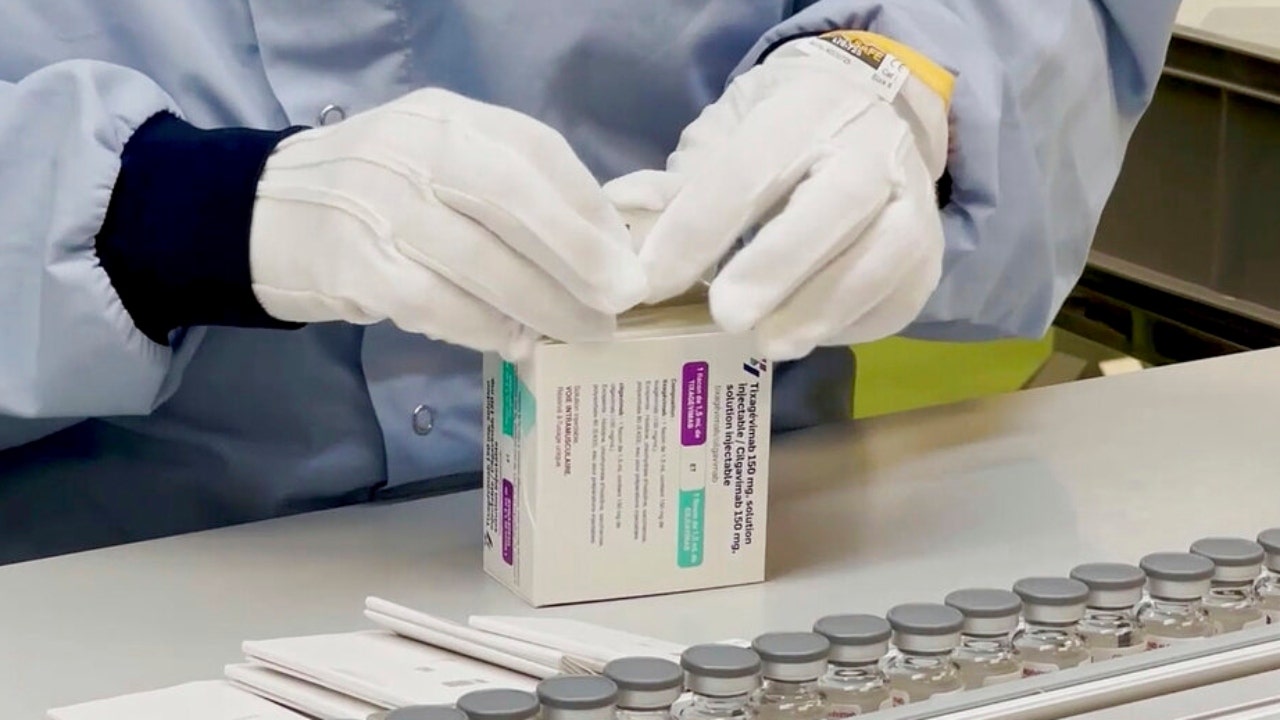
FDA authorizes AstraZeneca COVID antibody treatment for emergency use
Fox News
The U.S. Food and Drug Administration on Wednesday issued emergency use authorization for AstraZeneca's Evusheld.
In a news release, the agency said the authorization was issued for certain adults and individuals 12 years of age and older, weighing at least 40 kilograms (about 88 pounds).
Additionally, the product is only authorized for individuals who are not currently infected with COVID-19 and have not recently been exposed to someone who is.
More Related News

Pizza before tomatoes? Ancient Rome's version of America's favorite food looked nothing like today's
Ancient Rome pizza at Hungary restaurant features no tomatoes or mozzarella, using garum fish sauce and olive paste. Neverland Pizzeria's dish is limited-edition.












