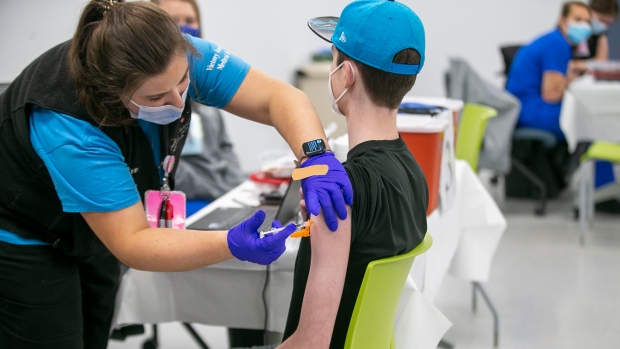
U.S. FDA paves way for Pfizer COVID-19 vaccinations in young kids
CTV
The U.S. Food and Drug Administration has paved the way for children ages 5 to 11 to get Pfizer's COVID-19 vaccine.
The FDA cleared kid-size doses -- just a third of the amount given to teens and adults -- for emergency use, and up to 28 million more American children could be eligible for vaccinations as early as next week.
One more regulatory hurdle remains: On Tuesday, advisers to the Centers for Disease Control and Prevention will make more detailed recommendations on which youngsters should get vaccinated, with a final decision by the agency's director expected shortly afterwards.
"Vaccinating younger children against COVID-19 will bring us closer to returning to a sense of normalcy," Dr. Janet Woodcock, the acting FDA commissioner, said in a statement. "Our comprehensive and rigorous evaluation of the data pertaining to the vaccine's safety and effectiveness should help assure parents and guardians that this vaccine meets our high standards."
A few countries have begun using other COVID-19 vaccines in children under 12, including China, which just began vaccinations for 3-year-olds. But many that use the vaccine made by Pfizer and its partner BioNTech are watching the U.S. decision, and European regulators just began considering the companies' kid-size doses.

While Canada is well known for its accomplishments in space — including building the robotic arms used on the Space Shuttle and the International Space Station — the country still has no ability to launch its own satellites. This week, Ottawa committed nearly a quarter‑billion dollars towards changing that.

It’s an enduring stereotype that Canadians are unfailingly nice, quick to apologize even when they have done nothing wrong. But an online urban legend claims the opposite of Canada’s soldiers, painting a picture of troops so brazen in their brutality that international laws were rewritten to rein them in.











