Natco Pharma’s generic Semaglutide set for roll out today
The Hindu
Natco Pharma will launch its Semaglutide injection (multi dose vials) in the domestic market on Saturday.
Natco Pharma will launch its Semaglutide injection (multi dose vials) in the domestic market on Saturday.
It will be available tomorrow, day 1 of patent expiry, the Hyderabad-based generic drugmaker said on Friday. Semaglutide is the active ingredient in drug major Novo Nordisk’s Ozempic and Wegovy. It is indicated for treatment of adults with type 2 diabetes as an adjunct to diet and exercise.
Last month, Natco had received Central Drugs Standard Control Organisation (CDSCO) approval to manufacture and market generic Semaglutide in India in multi dose vials and pen device based on the clinical comparison study.
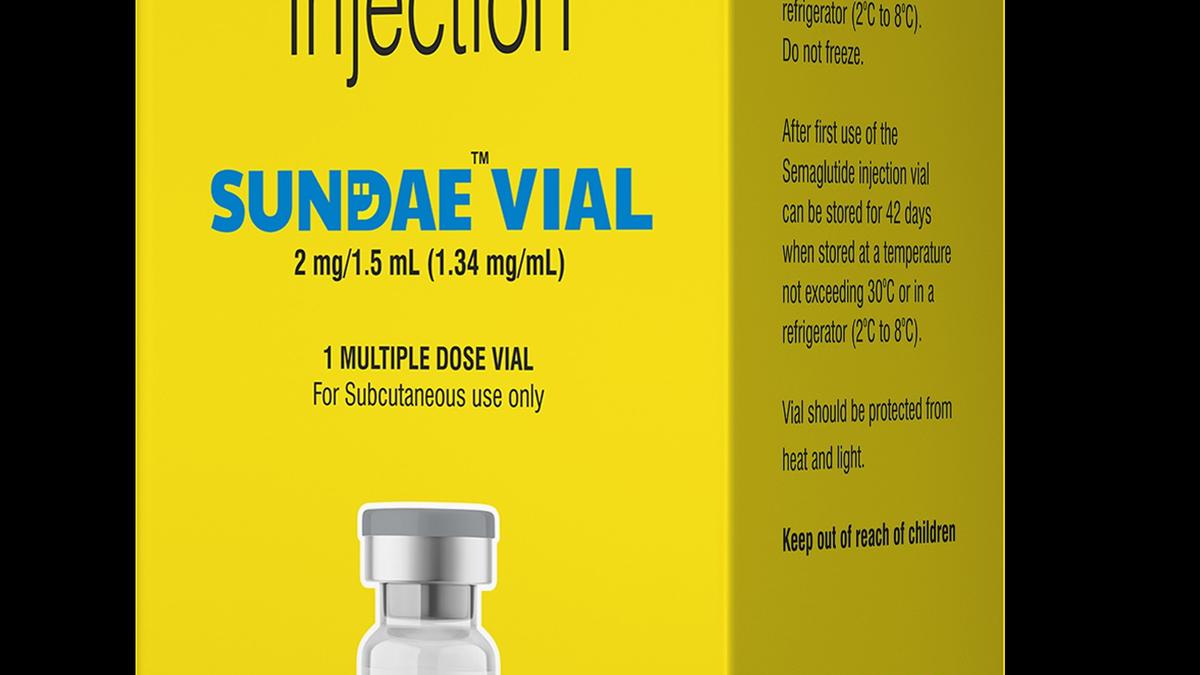
Semaglutide injection will be launched in multi dose vials of different strengths under brand names Semanat and Semanfull. The MRP of the vials will be ₹1,290 per month for 2 mg/1.5ml and 4mg/3ml; and ₹1,750 for 8mg/3ml.
The product in pen device is expected to be launched in April, in the price band of ₹4,000-4,500 per month. Natco said it is the first company to offer generic Semaglutide in multi dose vials with customised syringes. It is also offering the product to third parties for co-marketing.
Separately, Eris Lifesciences, which last month had announced a partnership with Natco for commercialisation of Semaglutide in the country, said it will launch the product under Sundae brand. A GLP-1 receptor agonist, Semaglutide has emerged as a transformative therapy for Type 2 diabetes and weight management, driven by strong outcomes in glycemic control and weight reduction, it said.













