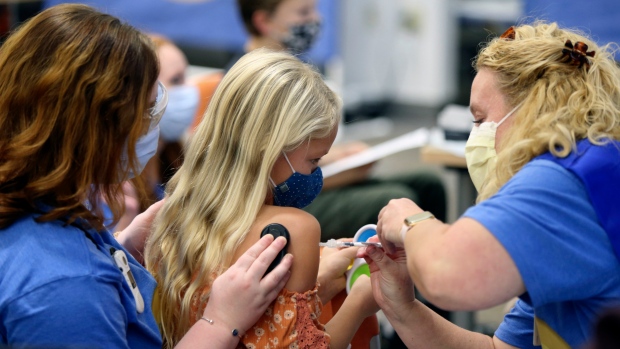
Canada could authorize vaccine for kids 5-11 in 'one to two weeks'
CTV
Health Canada says a decision on whether to authorize a COVID-19 vaccine for children will come 'in the next one to two weeks,' leaving open the possibility that some kids could be at least partially protected by Christmas.
The news comes as parts of Canada see an uptick in COVID-19 cases in the weeks leading up to the holiday season.
Canada's chief medical adviser Dr. Supriya Sharma said in a conference call with reporters Friday that the regulator is “actively continuing” its review of the Pfizer-BioNtech jab for children aged five to 11, which was authorized for use in the United States earlier this month.
Dr. Theresa Tam, Canada's chief medical officer, noted that the under-12 population - the only age group still ineligible for the vaccine - continues to have the highest incidence rates of COVID-19 across the country.
COVID-19 has typically shown to be mild in children since the pandemic began, but some kids have had serious illness. They can also spread the virus to others.

This year’s hard winter weather likely left significant damage for many homeowners coming into spring. Building and renovation expert Ryan Thompson spoke to CTV’s Your Morning about some of the biggest areas to focus on around the exterior of your home, to help prevent serious damage after the cold, hard winter.

While Canada is well known for its accomplishments in space — including building the robotic arms used on the Space Shuttle and the International Space Station — the country still has no ability to launch its own satellites. This week, Ottawa committed nearly a quarter‑billion dollars towards changing that.

It’s an enduring stereotype that Canadians are unfailingly nice, quick to apologize even when they have done nothing wrong. But an online urban legend claims the opposite of Canada’s soldiers, painting a picture of troops so brazen in their brutality that international laws were rewritten to rein them in.










