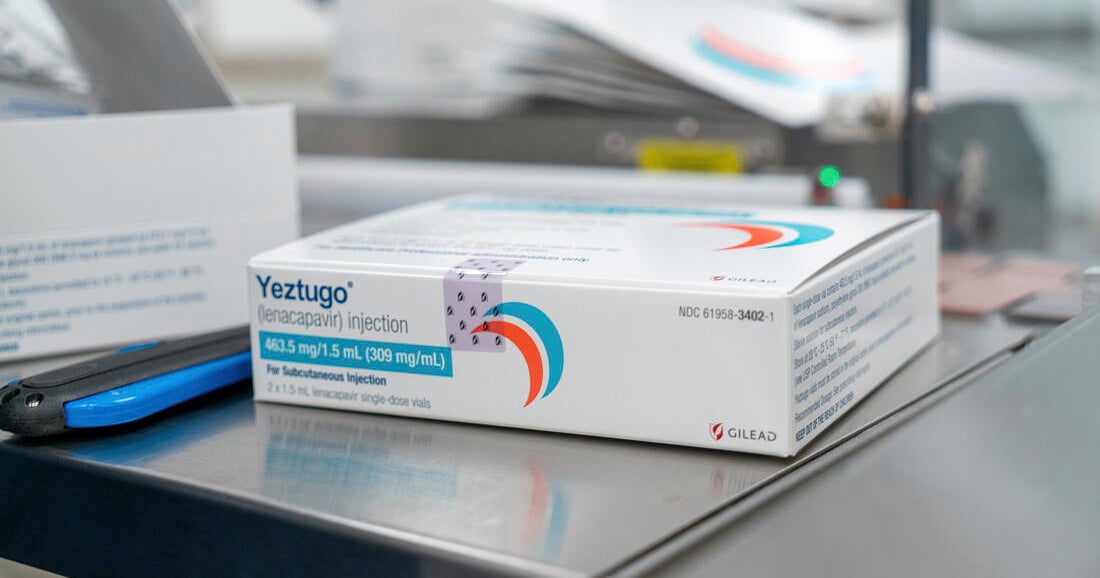
HIV prevention drug lenacapavir approved by FDA as twice-yearly injection
CBSN
The U.S. Food and Drug Administration has approved the drug lenacapavir as a twice-yearly injection to prevent HIV.
The drug, called Yeztugo from company Gilead Sciences, was approved Wednesday based on data from clinical trials that showed 99.9% of participants who received it remained HIV negative.
Daniel O'Day, Gilead's chairman and chief executive officer, called the approval a "milestone moment in the decades-long fight against HIV."

Washington — President Trump said early Monday that he is postponing airstrikes on Iran's power plants after "very good and productive conversations" over the last two days about reopening the Strait of Hormuz. However, Iran's foreign ministry denied any such talks. Claire Day contributed to this report. In:












