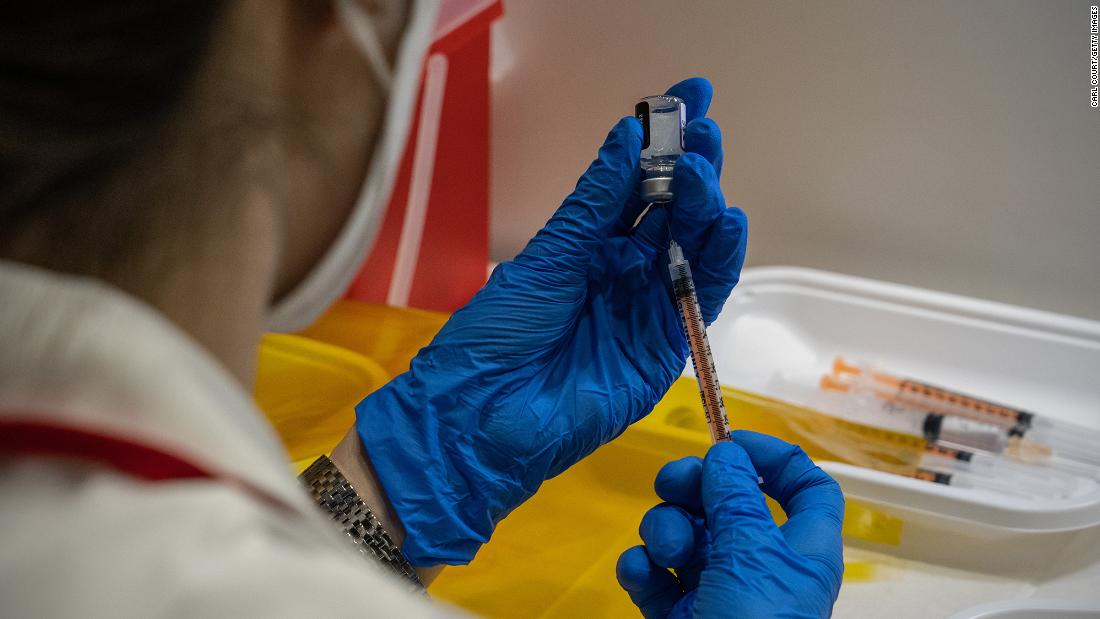
FDA postpones advisory panel meeting on Pfizer Covid-19 vaccine for younger children
CNN
The US Food and Drug Administration announced Friday that it is postponing the meeting of its Vaccines and Related Biological Products Advisory Committee, originally scheduled for February 15, as "new data have recently emerged" regarding Pfizer and BioNTech's emergency use authorization request for their Covid-19 vaccine for children younger than 5.
"This will give the agency time to consider the additional data, allowing for a transparent public discussion as part of our usual scientific and regulatory processes for COVID-19 vaccines. We will provide an update on timing for the advisory committee meeting once we receive additional data on a third dose in this age group from the company's ongoing clinical trial and have an opportunity to complete an updated evaluation," acting FDA Commissioner Dr. Janet Woodcock and Dr. Peter Marks, director of the FDA's Center for Biologics Evaluation and Research, said in a statement.
In a news release Friday, Pfizer said it continues to share data from its child vaccine trial with the FDA. With the surge of the Omicron variant, high rates of infection and illness among children have allowed the company to quickly accumulate the data it needs for the study protocol.

Janet Mills and her allies are counting on a gender gap to narrow Platner’s wide lead ahead of the June 9 primary to decide who will face incumbent Republican Sen. Susan Collins. They are betting that the unfiltered style that has brought Platner widespread attention as someone who could help Democrats reach young men will backfire with women.

As a shrinking number of Transportation Security Administration agents work to keep hourslong security lines moving despite not being paid, President Donald Trump stepped into the fray Saturday, announcing he will send Immigration and Customs Enforcement officers to airports by Monday if Congress doesn’t agree to a plan to end the partial government shutdown.











