FDA expands warning about potentially contaminated eye products amid outbreak
CBSN
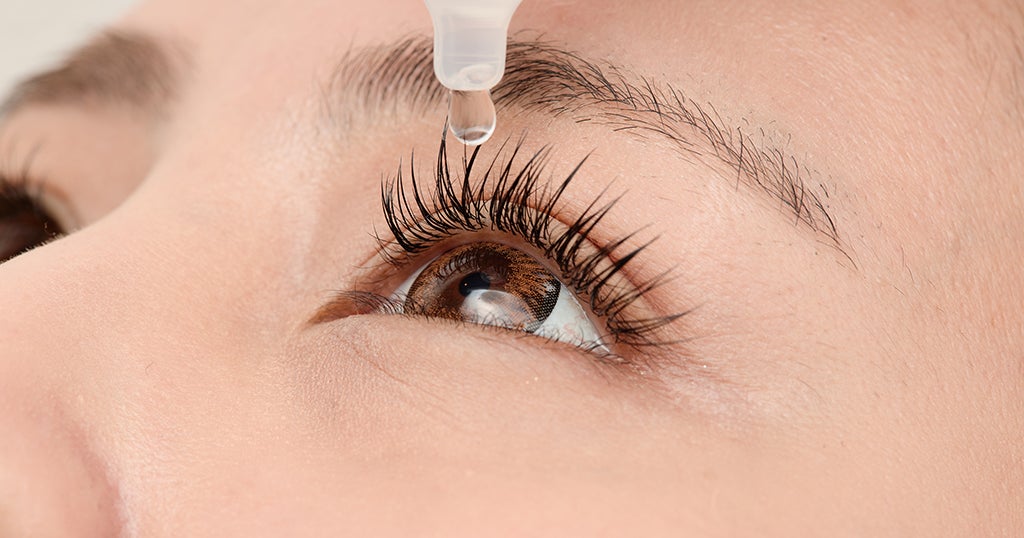
The Food and Drug Administration warned Wednesday of another imported eye product that could be contaminated with bacteria, in the wake of an outbreak earlier this year of highly drug-resistant bacteria that hospitalized and blinded some patients.
Americans should stop using Delsam Pharma's Artificial Eye Ointment, the FDA says, an over-the-counter product manufactured by Global Pharma Healthcare Private Limited.
It's the same company behind the EzriCare and Delsam Pharma artificial tears products that were recalled earlier this year. The FDA had faulted the company for multiple violations and banned imports into the United States of its items.

The Federal Communication Commission announced Thursday evening that it had approved the $6.2 billion merger of major broadcast station owners Nexstar and Tegna. The move came on the same day that attorneys general in eight states and DirecTV filed separate lawsuits seeking to block the deal, arguing that it will lead to higher prices for consumers and stifle local journalism. In:












