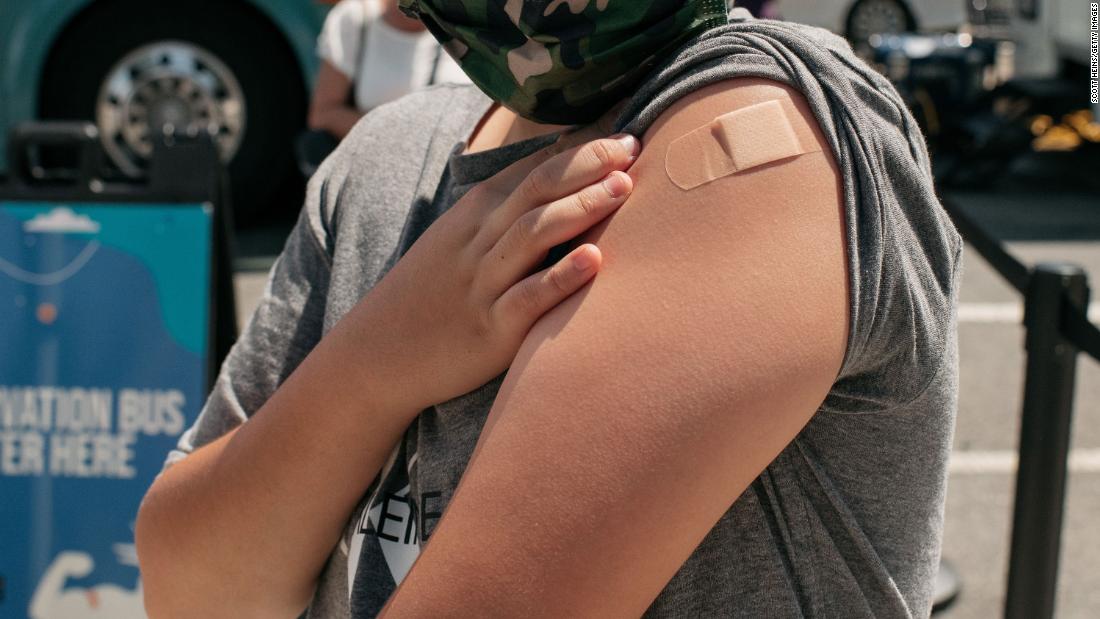
FDA expands booster eligibility to adolescents and shortens time needed after primary series
CNN
The US Food and Drug Administration on Monday expanded the emergency use authorization for Pfizer's Covid-19 vaccine boosters to children ages 12 to 15.
The agency also shortened the time needed before receiving a booster shot from at least six months after completion of the initial series to at least five months, for everyone 12 and older.
"Throughout the pandemic, as the virus that causes Covid-19 has continuously evolved, the need for the FDA to quickly adapt has meant using the best available science to make informed decisions with the health and safety of the American public in mind," FDA Acting Commissioner Dr. Janet Woodcock said in a statement. "With the current wave of the omicron variant, it's critical that we continue to take effective, life-saving preventative measures such as primary vaccination and boosters, mask wearing and social distancing to in order to effectively fight Covid-19."

Oklahoma’s governor picks energy executive Alan Armstrong to fill US Senate seat through end of year
Oklahoma’s governor on Tuesday appointed energy executive Alan Armstrong to serve in the US Senate through the end of the year and finish the term of Republican Markwayne Mullin, the new homeland security secretary.












